What We Do
Research
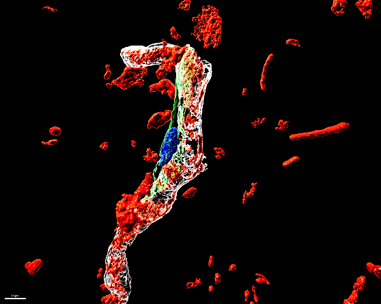
Role of blood-derived coagulation factors in orchestrating glial and fibrotic scar formation
CNS injuries and diseases are associated with chronic non-resolving scar formation that hinders brain repair. We identified the circulating blood factor fibrinogen, which is not synthesized within the brain, but leaks from the bloodstream into the CNS parenchyma after vascular damage or BBB disruption, as the initial astrocyte activation signal. Fibrinogen deposition at scar borders drives astrocyte morphology and inhibitory ECM deposition. Furthermore, fibrinogen is enriched in stem cell niches regulating the misguided differentiation of adult neural stem/precursor cells into reactive astrocytes that contribute to the inhibitory scar. Currently, we investigate how circulating blood-derived factors regulate the border formation between the glial scar and the non-neural fibrotic scar in CNS disease. The identified mechanisms are the basis for manipulating peripheral factor - scar forming cell interactions to resolve pathological scar formation in the CNS.
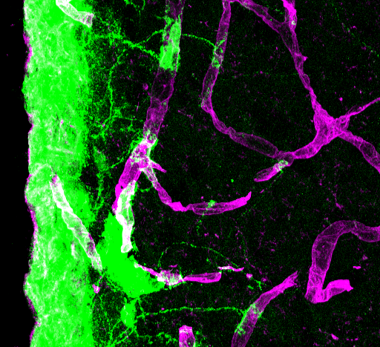
Harnessing neural stem cells for improving brain repair
Our current results highlight the functional significance of endogenous neural stem cells (NSCs) as signal mediators between the CNS and the peripheral immune system, and uncover their potential as therapeutic targets to modulate structural and immune cells for improving brain repair. Currently, we are testing the effects of modified endogenous NSCs and modified transplanted human induced pluripotent stem cell (iPSC)-derived NSCs and glial precursor cells on immunmodulation, scar formation and functional regeneration.
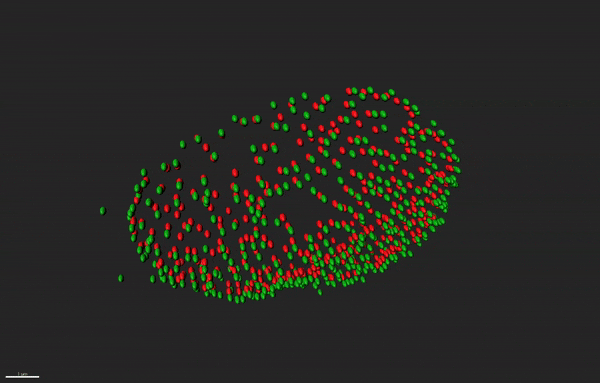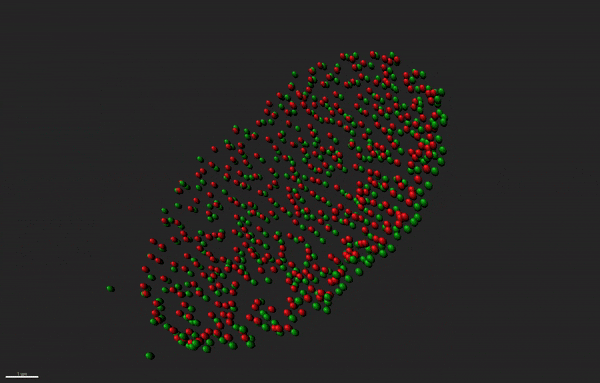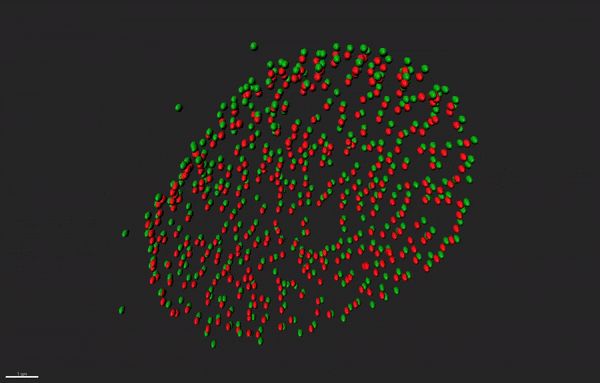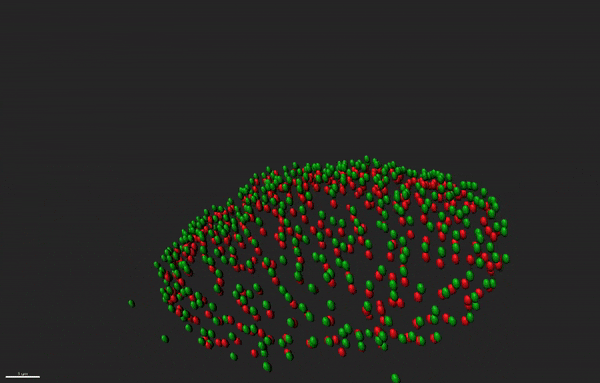
Super resolution microscopy of nuclear pore complex remodeling in NSCs
Nuclear pore complexes (NPC) are highly dynamic macromolecular protein structures that facilitate molecular exchange across the nuclear envelope. Aberrant NPC functioning has been implicated in neurodegeneration. With the aid of super resolution microscopy, we study how changes in the functions of NPC constituents, the nucleoporins, may impair neurogenesis in neurodegenerative diseases, such as Alzheimer’s disease.

